Research Projects

Nitrogen fixation in the North Atlantic Gyre
Reactive nitrogen required for phytoplankton growth is introduced to the biosphere by biological nitrogen fixation, the conversion of dinitrogen gas (N2) to biologically available ammonia (NH3). In the ocean, this fundamental reaction is performed by unicellular
prokaryotic plankton, including cyanobacteria and some heterotrophic bacteria. In spite of years of active research on oceanic N2 fixation, the factors that govern where, when and why plankton fix N2 remain open questions. The subtropical North Atlantic gyre is considered to be a globally significant hot spot for biological N2 fixation, although the region’s nutrient reservoir below the surface is very enriched in nitrate relative to nutrient phosphate – raising questions as to how N2 fixation is sustained in the gyre. In collaboration with Drs. Jamie Palter (URI), Pia Moisander (UMass Dartmouth) and Angel White (OSU), and Mar Benavides (Mediterranean Institute of Oceanography, France) we are investigating the hypothesis that nutrient phosphate in excess of nitrate entrained in the North Atlantic Gulf Stream fuel substantial rates of biological N2 fixation in the gyre boundary. During a recent expedition across the Atlantic Gulf Stream aboard the R.V. Endeavor, we performed on-deck incubations to quantify N2 fixation in surface and subsurface waters, paired with hydrographic measurements and a concurrent investigation of N2 fixer abundances and of the presence of nitrogenase genes.
Limit of detection of N2 fixation measurements
Biological N2 fixation is a dominant source reactive N to the biosphere – yet a poorly constrained term of the global N cycle. In the ocean, N2 fixation is typically detected at surface of tropical ocean, effectuated by diazotrophic cyanobacteria. Recent studies, however, report the detection of relatively low N2 fixation rates in the dark mesopelagic ocean. Extrapolation of these rates to the large volume of the meso- to bathypelagic waters suggests that N2 fixation in the deep ocean could potentially contribute a substantial quantity of new reactive N to the system. However, reported rates are exceedingly low, raising questions as to minimum quantifiable rates of the 15N2 tracer method in oceanic systems. Moreover, while DNA-based surveys report the presence of non-cyanobacterial nifH sequences in aphotic layers, detected gene copy abundances appear insufficient to justify measured rates and evidence for expression of these genes in the mesopelagic and below is quasi non-existent
In collaboration with Pia Moisander, Angel White, and Mar Benavides, we are conducting a thorough examination of potential experimental and analytical errors inherent to the 15N2-tracer N2 fixation method, in tandem with comprehensive molecular measurements in the deep ocean waters, in order to (1) determine the minimum quantifiable rates of 15N2 fixation based on incubations of mesopelagic waters via full characterization of sources of experimental error and mass spectrometer performance, and (2) seek evidence of expression and presence of nitrogen fixation genes via comprehensive molecular approaches on corresponding samples.
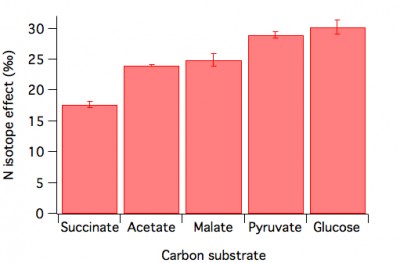
Controls on the denitrification isotope effect (NSF-OCE)
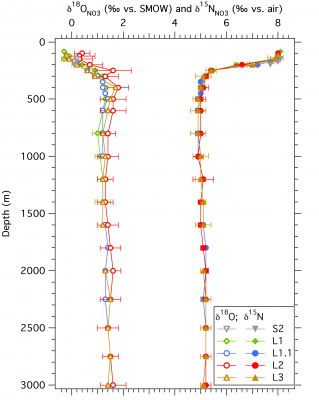
GEOTRACES: N biogeochemistry of the western Arctic Ocean inferred from nitrate N and O isotope ratios (NSF-OCE/PLR)
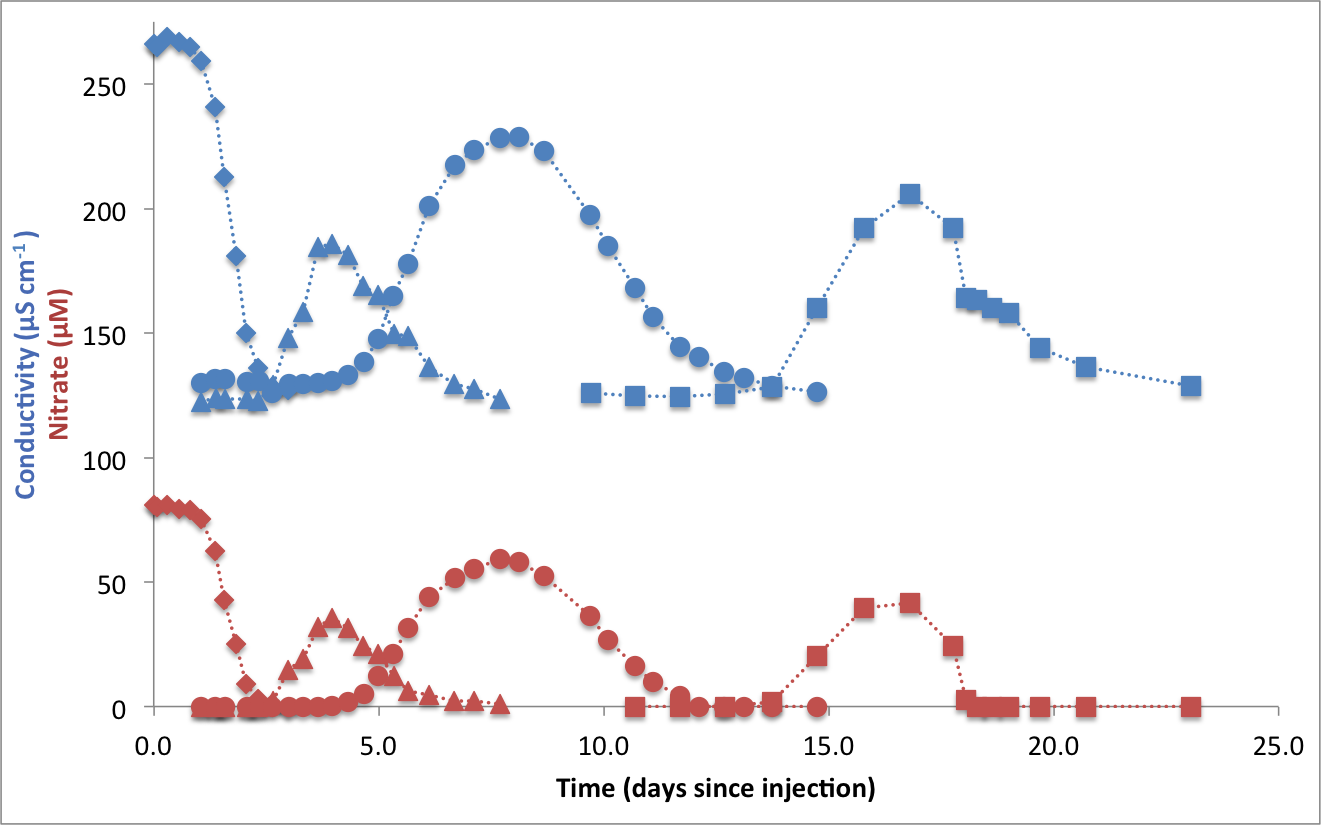
Nitrate isotope dynamics in groundwater aquifers (NSF-EAR)
aka. The Cape Cod Aquifer Project. Collaborators: Craig Tobias, J. K. Böhlke, and Richard Smith
In June this year (2016), we injected nitrate and acetate in the anoxic portion of a USGS experimental aquifer in Cape Cod (http://toxics.usgs.gov/), in order to monitor the evolution of nitrate N and O isotope ratios when denitrification by heterotrophic bacteria is stimulated (by acetate) over oxidative nitrate production (by anammox and/or nitrification). To do so, we first pumped aquifer-water to the surface into a steel drum containing nitrate, acetate and bromide (a conductivity tracer), whilst keeping groundwater anoxic, then the mixture was pumped back into the ground, and the evolution of the nitrate plume and its isotope was tracked down-gradient for a month. Graduate student Danielle Boshers will be measuring the N and O isotopes of nitrate and nitrite from the collected samples this summer.
N-isotope cycling in the California Upwelling
collaboration with Dr. Jason Smith
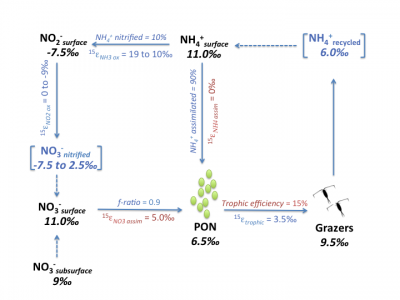 Jason has visited the Granger lab to make measurements 15N/14N ratios of nitrate, nitrite, ammoniu, PON and zooplankton grazers in the California Upwelling. These measurements (black text) enable to assess new vs. recycled N uptake by phytoplankton, as well as the fraction of ammonium that is nitrified to nitate vs. assimilated, given assumptions (red text) on some isotope effects and grazer transfer efficency.
Jason has visited the Granger lab to make measurements 15N/14N ratios of nitrate, nitrite, ammoniu, PON and zooplankton grazers in the California Upwelling. These measurements (black text) enable to assess new vs. recycled N uptake by phytoplankton, as well as the fraction of ammonium that is nitrified to nitate vs. assimilated, given assumptions (red text) on some isotope effects and grazer transfer efficency.